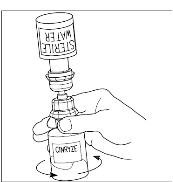
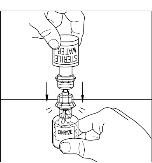
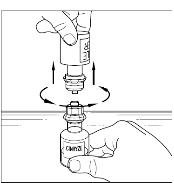
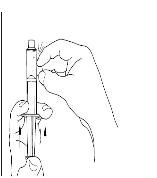
Final Results from the Longest Hereditary Angioedema Study of Active Treatment Duration Conducted to Date Support the Sustained Safety and Efficacy of TAKHZYRO® (lanadelumab) Injection for Long-Term Prevention of Hereditary Angioedema Attacks

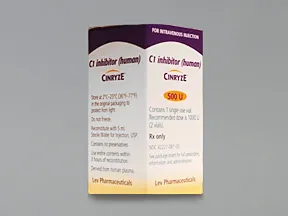
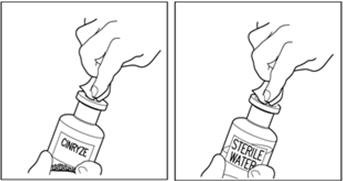
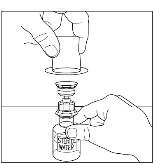
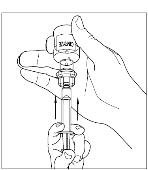
Takeda launches CINRYZE™ in India, the first C1-I NH for prophylaxis in hereditary angioedema patients

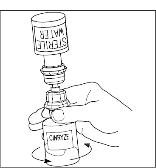
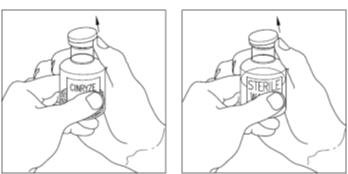
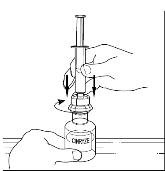
Successful Long-Term Prophylactic Treatment With Subcutaneous C1 Esterase Inhibitor in a Patient With Hereditary Angioedema - Janina Hahn, Melanie Nordmann-Kleiner, Susanne Trainotti, Thomas K. Hoffmann, Jens Greve, 2020

![CINRYZE® (C1 esterase inhibitor [human]) for Hereditary Angioedema CINRYZE® (C1 esterase inhibitor [human]) for Hereditary Angioedema](https://www.cinryze.com/dist/images/hdr-logo2.webp)